Walton Family Pharma Hub - Page 2

State and federal courts have wildly different rules for replacing lawyers during a case. Getting it wrong can cost you your case, your client, or your license. Here’s what you need to know before you file a substitution motion.
- Read More

Regulatory exclusivity provides government-granted market protection for pharmaceuticals beyond patents. It ensures innovators have commercial time even if patents expire during development. Key types include 5-year NCE, 12-year biologics, and 7-year orphan drug exclusivity. Differences exist between U.S. and EU rules, with significant economic impacts on drug pricing and competition.
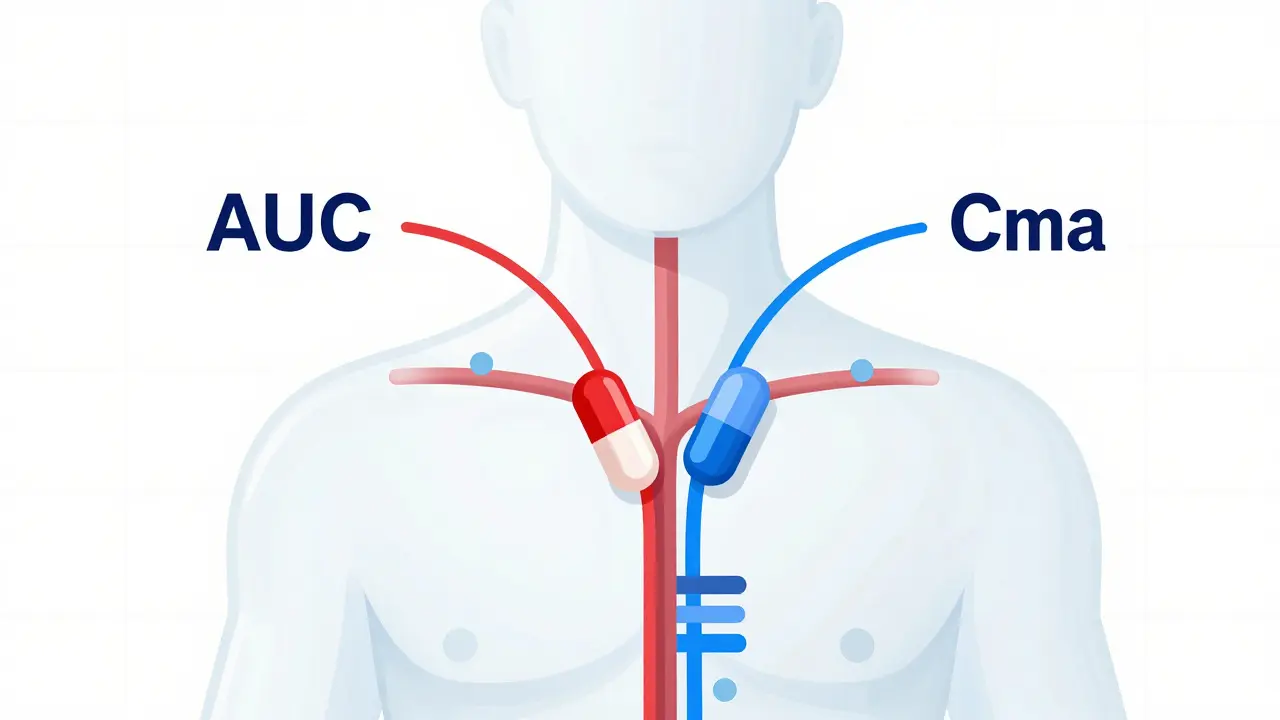
Generic Absorption Rates: How They Must Match Brand Drugs to Ensure Safety and Effectiveness
- Feb, 3 2026
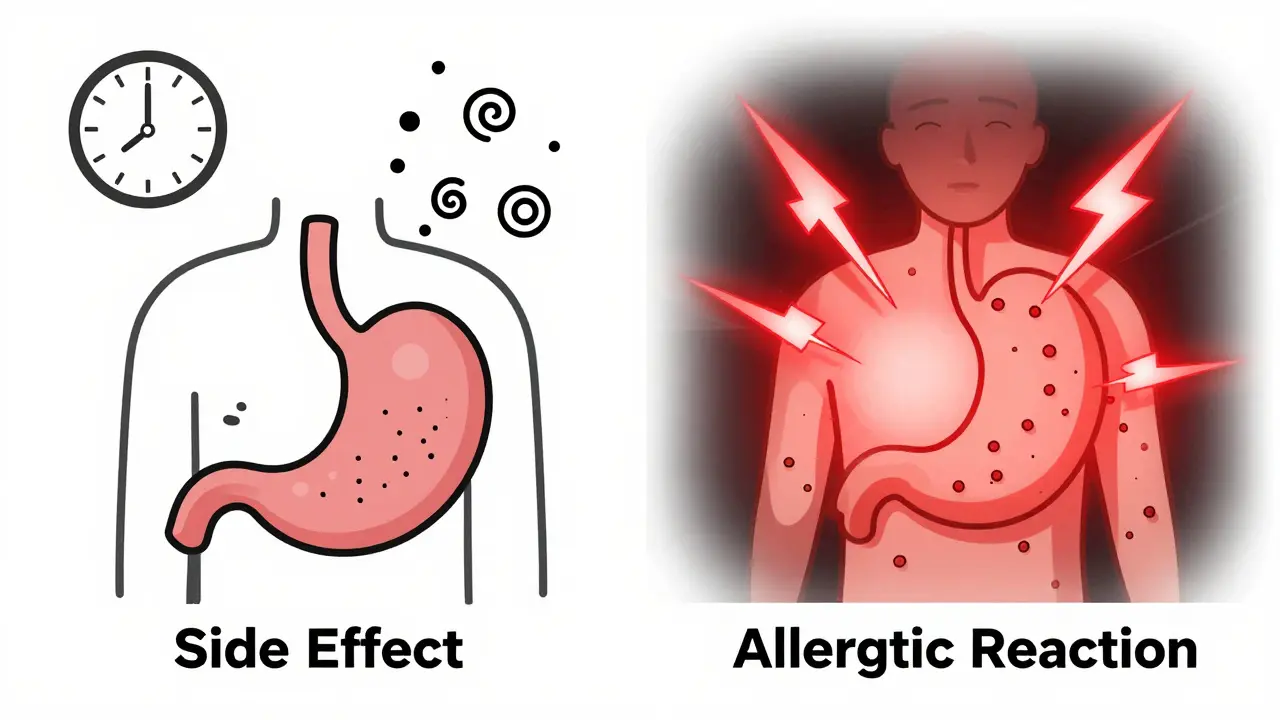
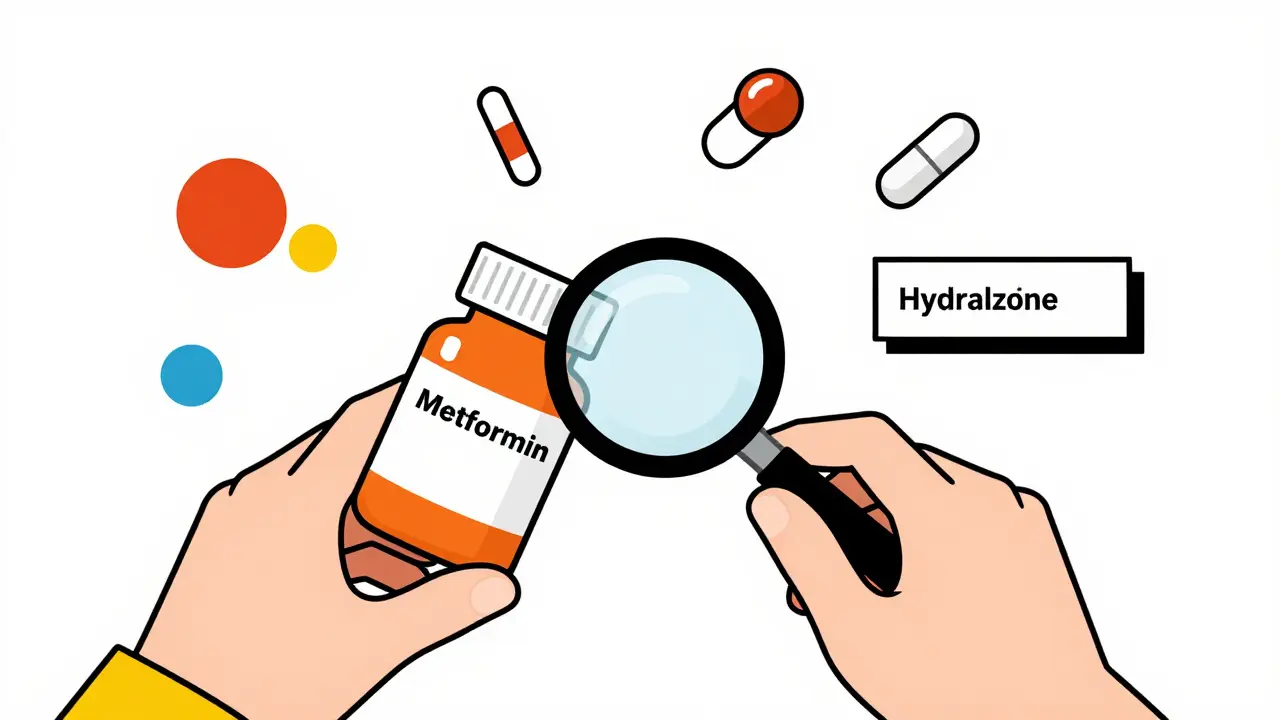
Five Essential Rules for Taking Medication Safely
- Jan, 28 2026